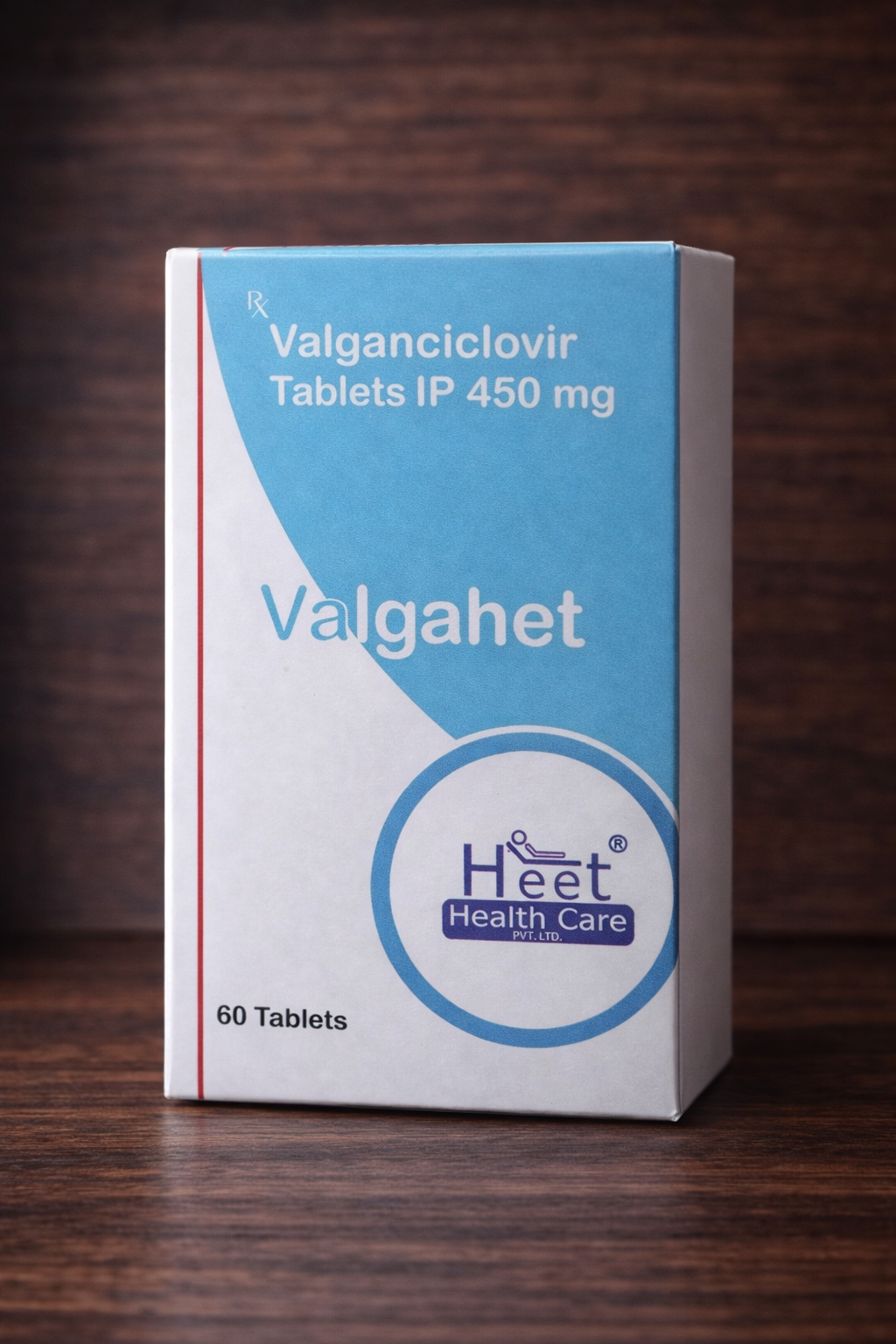
Valgahet (Valganciclovir) 450 mg Tablets №60
€145,00
Valganciclovir 450 mg is a prescription systemic antiviral medication used as prescribed by a physician for the treatment and prevention of cytomegalovirus (CMV) infection in at-risk patients, including those with immunodeficiency conditions and after transplantation. On iDoc.biz, you can check availability, packaging, delivery conditions, and receive consultation before placing an order.
Key Information
Active substance: valganciclovir
Dosage: 450 mg
Dosage form: tablets
Pack size: No. 60
Pharmacological group: systemic antiviral agent
Main indication: treatment and prevention of CMV infection as prescribed by a physician
Monitoring during therapy: blood tests, kidney function
Pregnancy and planning: requires mandatory medical consultation
Self-medication: not allowed
.
Description
Valganciclovir
is a prescription systemic antiviral medication containing the active substance valganciclovir. It is used as prescribed by a physician for the treatment and prevention of cytomegalovirus (CMV) infection in patients at risk, including those with immunodeficiency conditions and in certain cases after transplantation. Valganciclovir belongs to therapies that require medical supervision, so it is important to consult a healthcare professional before ordering and using it.
After oral administration, valganciclovir is converted into its active metabolite ganciclovir and helps suppress viral DNA replication. In practice, this means the drug is used as part of antiviral therapy for CMV infection when treatment must be carried out under medical supervision. Valganciclovir is not intended for self-selection of treatment regimens and should not be used without medical indication.
What valganciclovir is used for
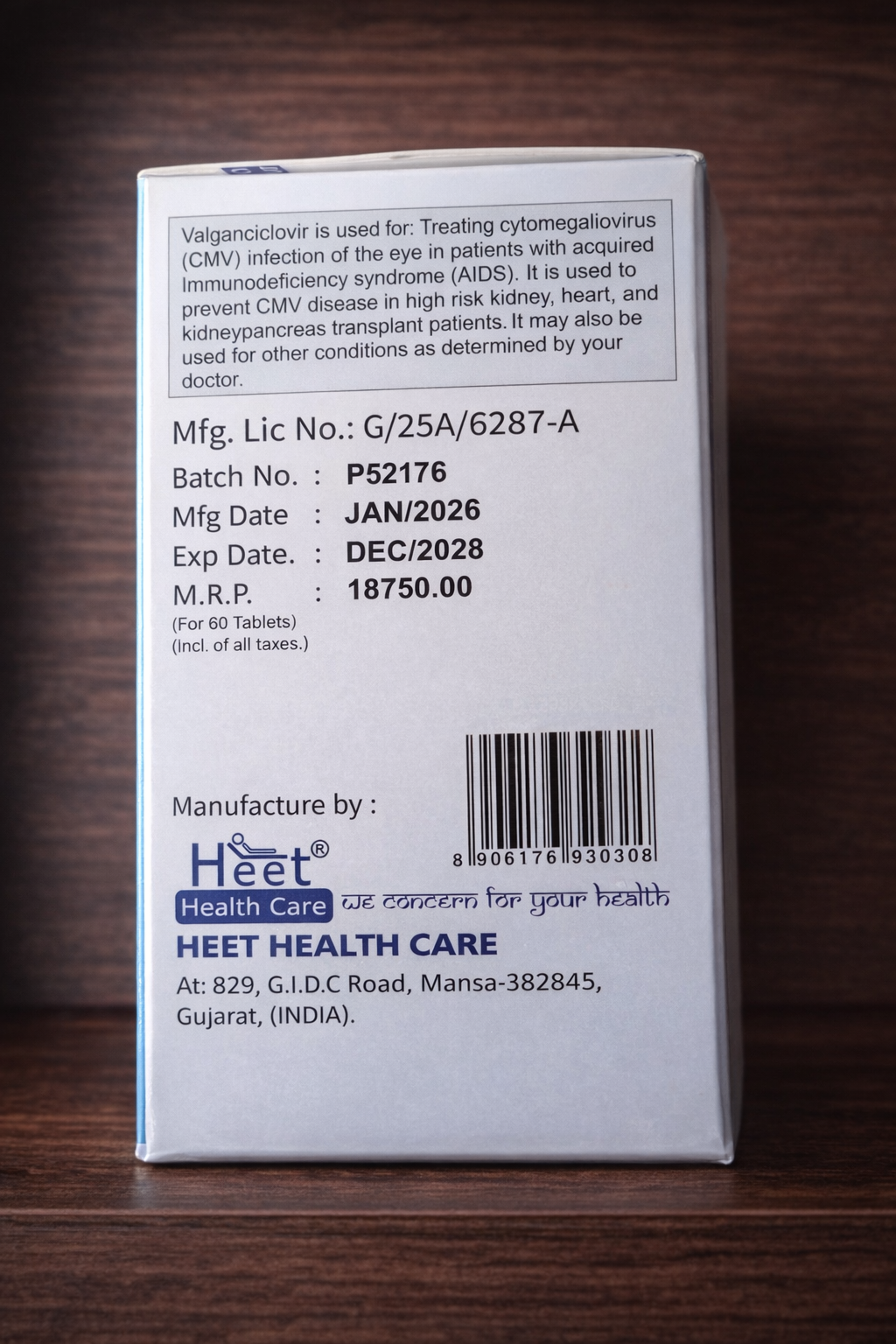
The drug is prescribed by a physician in clinical situations associated with cytomegalovirus infection. According to official patient sources, valganciclovir is used to treat CMV retinitis in certain patient groups, as well as to prevent CMV disease in some patients after transplantation. Specific indications, duration of therapy, and choice of dosage form are determined by the treating physician, taking into account the diagnosis, kidney function, concomitant therapy, and the patient’s overall condition.
Important information before use
Valganciclovir is a medication that requires special caution. During therapy, a physician may prescribe regular monitoring of complete blood count and kidney function, as the drug is associated with the risk of decreased red blood cells, white blood cells, and platelets, as well as renal complications. In addition, official sources highlight risks during pregnancy, potential effects on fertility, and the need for caution when combined with certain other medications. Self-medication with this drug is not acceptable.
What to tell your doctor
Before starting therapy, patients should inform their doctor about any kidney or liver disease, blood disorders, pregnancy, plans for pregnancy, breastfeeding, and all medications being taken, including prescription drugs, over-the-counter medicines, vitamins, dietary supplements, and immunosuppressants. This is especially important because valganciclovir may interact with other medications and may require adjustments in monitoring or treatment.
How to use
Tablets should be taken orally as prescribed by a physician. Official information emphasizes that tablets should be swallowed whole and used only as directed by a healthcare professional. Do not change the dose, frequency, or discontinue treatment without consulting your doctor, as this may affect treatment effectiveness and increase the risk of adverse reactions.
Possible side effects
Adverse reactions may occur during therapy, including nausea, diarrhea, vomiting, headache, fatigue, blood-related disorders, and other side effects. If symptoms such as severe weakness, pallor, unusual bleeding or bruising, signs of infection, vision changes, decreased urination, seizures, or other serious symptoms occur, medical attention should be sought immediately.
Storage conditions
The tablet form is usually stored at room temperature, protected from excessive heat and moisture, and kept out of reach of children. Do not use the product after the expiration date. Any additional storage conditions should follow the manufacturer’s instructions and packaging information.
Ordering on iDoc.biz
On iDoc.biz, you can check the availability of the required package, ask questions about ordering conditions, and receive consultation before placing an order. Support is available daily from 09:00 to 18:00 (Kyiv time), and responses in messengers usually take 5–10 minutes. Before ordering, we recommend уточнить the current packaging, manufacturer, country of origin, and available delivery conditions.
FAQ
What is valganciclovir used for?
Valganciclovir is used as prescribed by a physician in clinical situations related to cytomegalovirus infection, including treatment of CMV retinitis in certain patients and prevention of CMV disease in some patients after transplantation.
Can valganciclovir be purchased without a prescription?
It is a prescription medication and should only be used under medical supervision.
Is monitoring required during treatment?
Yes, doctors may require monitoring of blood counts and kidney function due to the risk of serious side effects.
Can it be used during pregnancy?
Use during pregnancy must be discussed with a physician. Self-medication is not acceptable.
Can the dose be changed or treatment stopped independently?
No. The treatment regimen is determined by a physician, and самостоятельные изменения могут быть небезопасны.
How should tablets be stored?
Tablets should be stored at room temperature, protected from moisture and excessive heat, and kept out of reach of children. Refer to the packaging of the specific manufacturer for details.
How to check availability and ordering conditions on iDoc.biz?
You can contact us via WhatsApp, Viber, Telegram, or email. iDoc.biz support is available daily from 09:00 to 18:00 (Kyiv time).
The information on this page is for reference purposes only and does not replace medical advice. The product should be used only under medical supervision and based on appropriate medical indications.
Additional information
| Djelatna tvar | valganciklovir |
|---|---|
| Farmaceutski oblik | tablete |
| Doza | 450 mg |
| Pakiranje | 60 tableta |
| Zemlja proizvodnje: | Indija |





Reviews
There are no reviews yet.