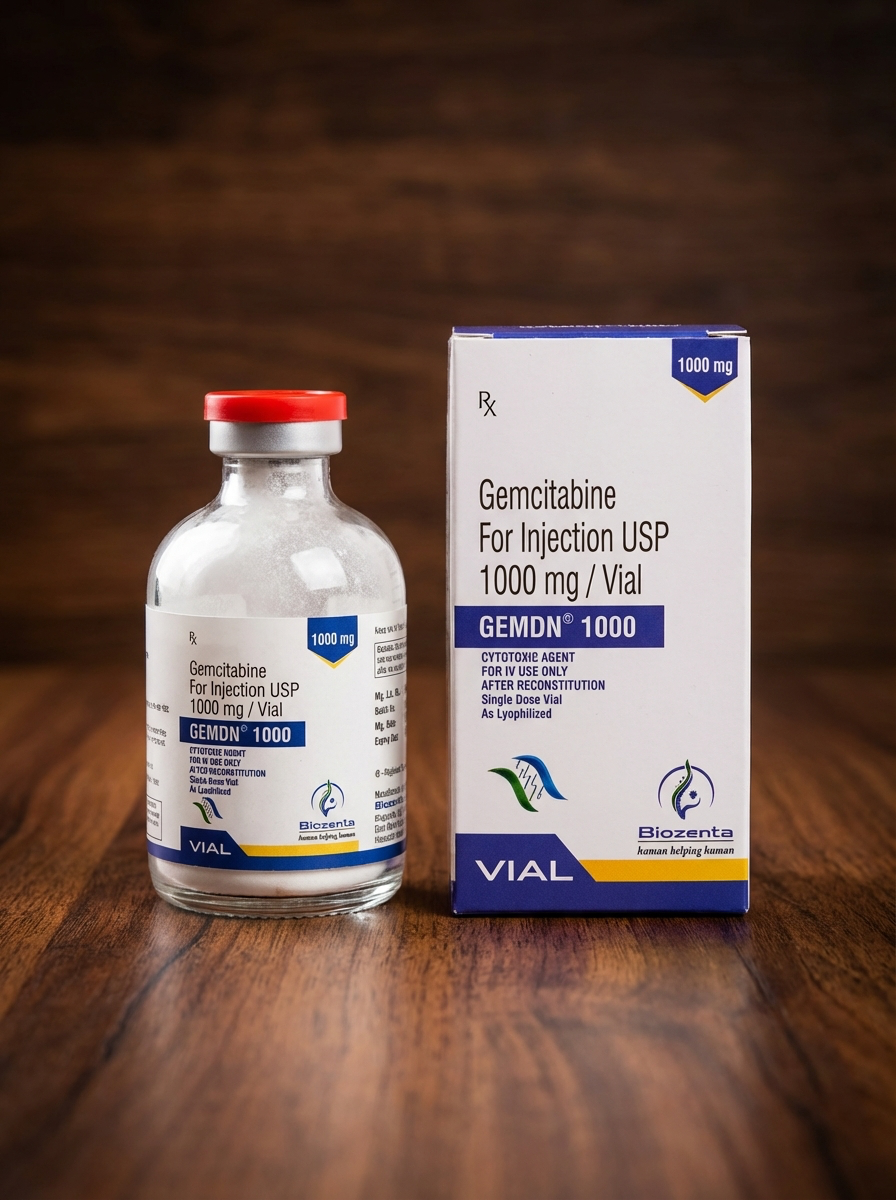
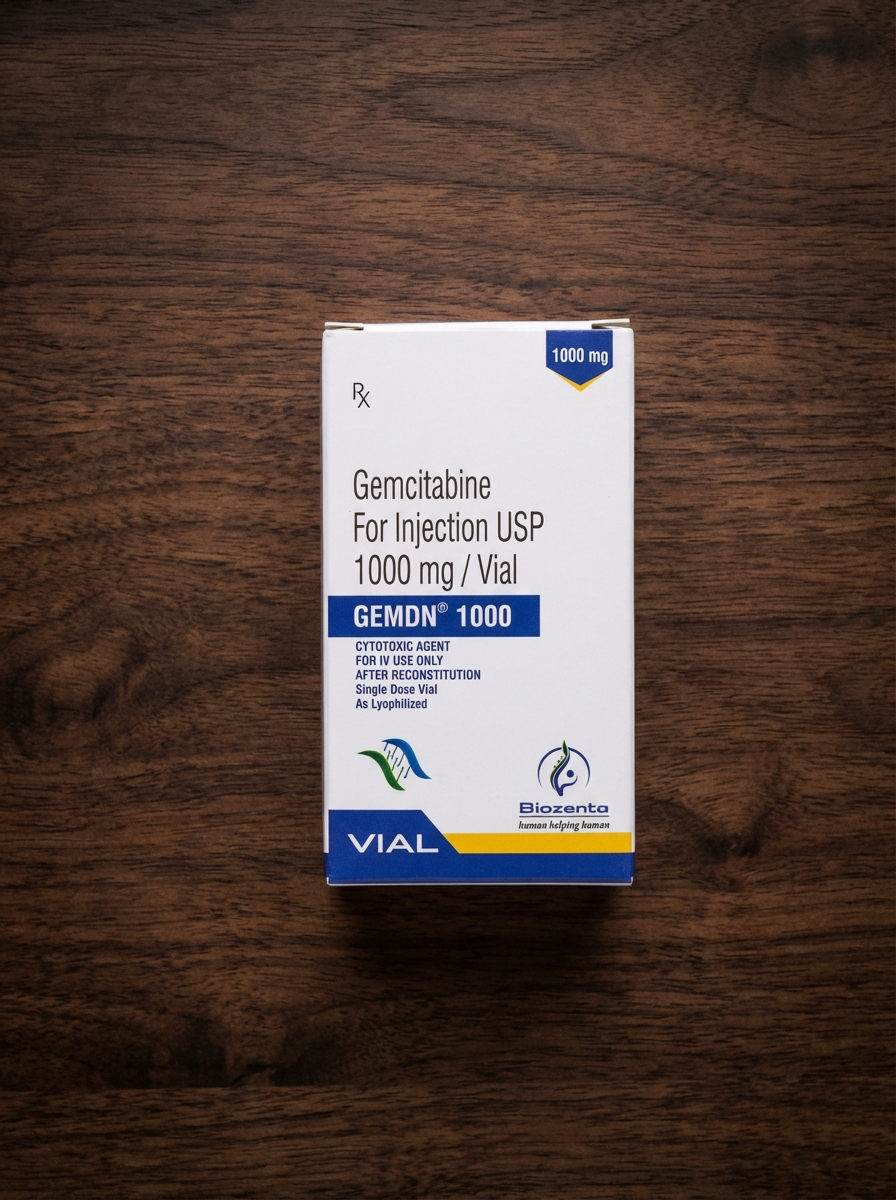
Gemdn 1000 (Gemcitabine) Powder for Injection 1000 mg
€33,00
Gemdn 1000 (gemcitabine) is a prescription cytotoxic anticancer medication from the antimetabolite class used in chemotherapy for various malignant tumors. It inhibits DNA synthesis in tumor cells, slowing their growth and division. Used only under medical supervision.
Description
Gemdn 1000 (gemcitabine) is a prescription cytotoxic antineoplastic medication used in systemic oncology therapy for the treatment of various malignant tumors. It belongs to the class of antimetabolites (pyrimidine analogs) and is administered intravenously under strict medical supervision. The drug is widely used in modern chemotherapy protocols and is considered an effective component of combination anticancer regimens.
Pharmacological Class
Antineoplastic agent — antimetabolite cytotoxic drug.
Mechanism of Action
Gemcitabine enters tumor cells and is converted into active metabolites that integrate into DNA during synthesis. This process:
-
inhibits DNA synthesis
-
blocks cellular replication
-
stops tumor cell division
-
triggers programmed cell death (apoptosis)
As a result, tumor growth slows and cancer progression may be controlled.
Indications
Gemdn 1000 is used in oncology for treatment of:
-
pancreatic cancer
-
non-small cell lung cancer (NSCLC)
-
breast cancer
-
other solid tumors as part of combination chemotherapy
The exact treatment regimen is determined individually by an oncologist.
Dosage Form
-
Lyophilized powder for solution for infusion
-
Strength: 1000 mg
-
Pack size: 1 vial
Administration
The drug is administered intravenously after reconstitution. Dose, frequency, and duration of treatment depend on:
-
tumor type and stage
-
patient body surface area
-
previous therapies
-
overall patient condition
Treatment is usually given in cycles with rest periods between courses to allow recovery.
Monitoring During Therapy
Regular medical monitoring is required, including:
-
complete blood count
-
liver and kidney function tests
-
infection surveillance
-
clinical condition assessment
This is essential due to possible bone marrow suppression and systemic toxicity.
Possible Side Effects
Potential adverse reactions include:
-
neutropenia, thrombocytopenia, anemia
-
nausea and vomiting
-
fatigue and weakness
-
skin reactions
-
increased risk of infections
Severe symptoms require immediate medical attention.
Contraindications
-
hypersensitivity to gemcitabine
-
severe bone marrow suppression
-
pregnancy and breastfeeding without medical supervision
Drug Interactions
Patients should inform their physician about all medications they are taking, as gemcitabine may interact with other drugs, especially those affecting blood formation or immune function.
Special Precautions
-
Treatment must be supervised by an oncologist.
-
Self-medication is not permitted.
-
Dose adjustments may be required depending on laboratory results and tolerability.
Pregnancy and Fertility
Gemcitabine may harm a developing fetus. Effective contraception is recommended during therapy.
Prescription Status
Prescription only.
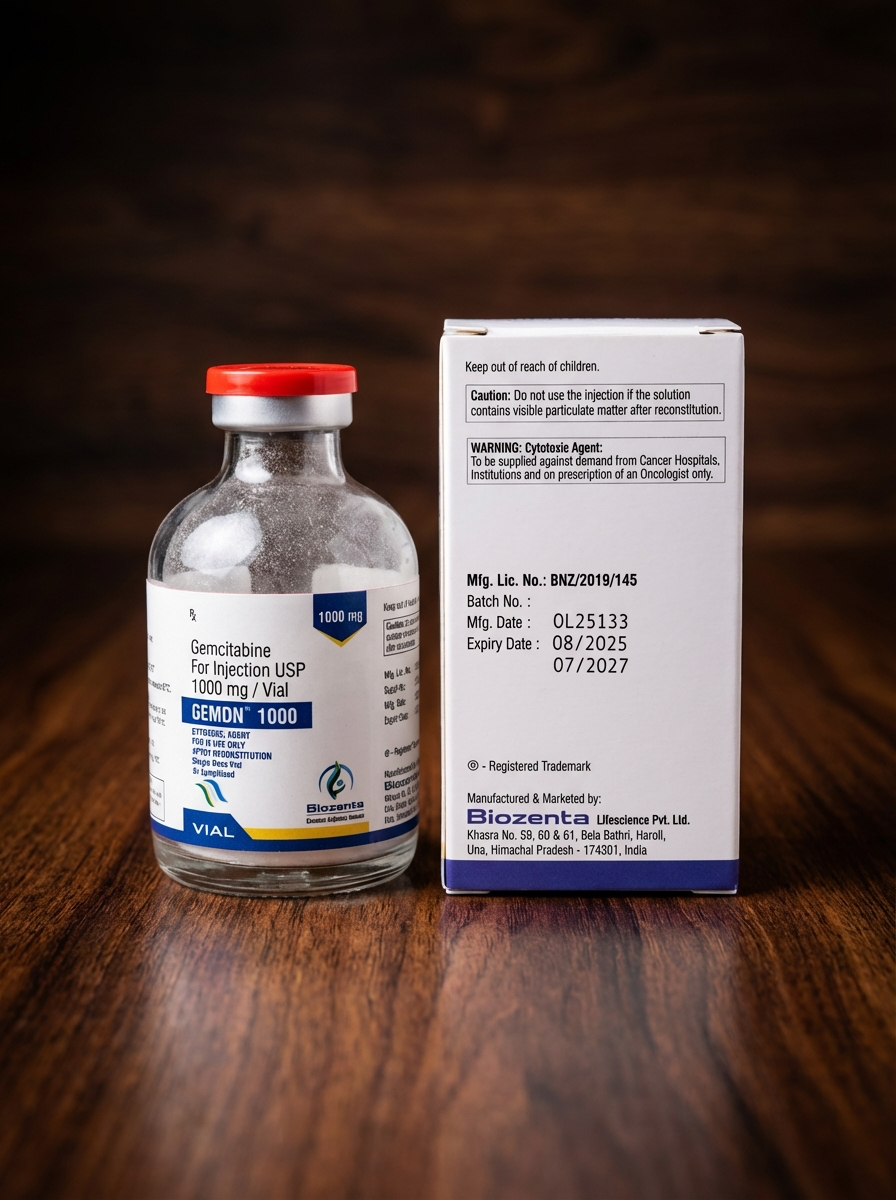
Manufacturer
Biozenta Lifescience Pvt. Ltd.
Country of Manufacture
India


Reviews
There are no reviews yet.