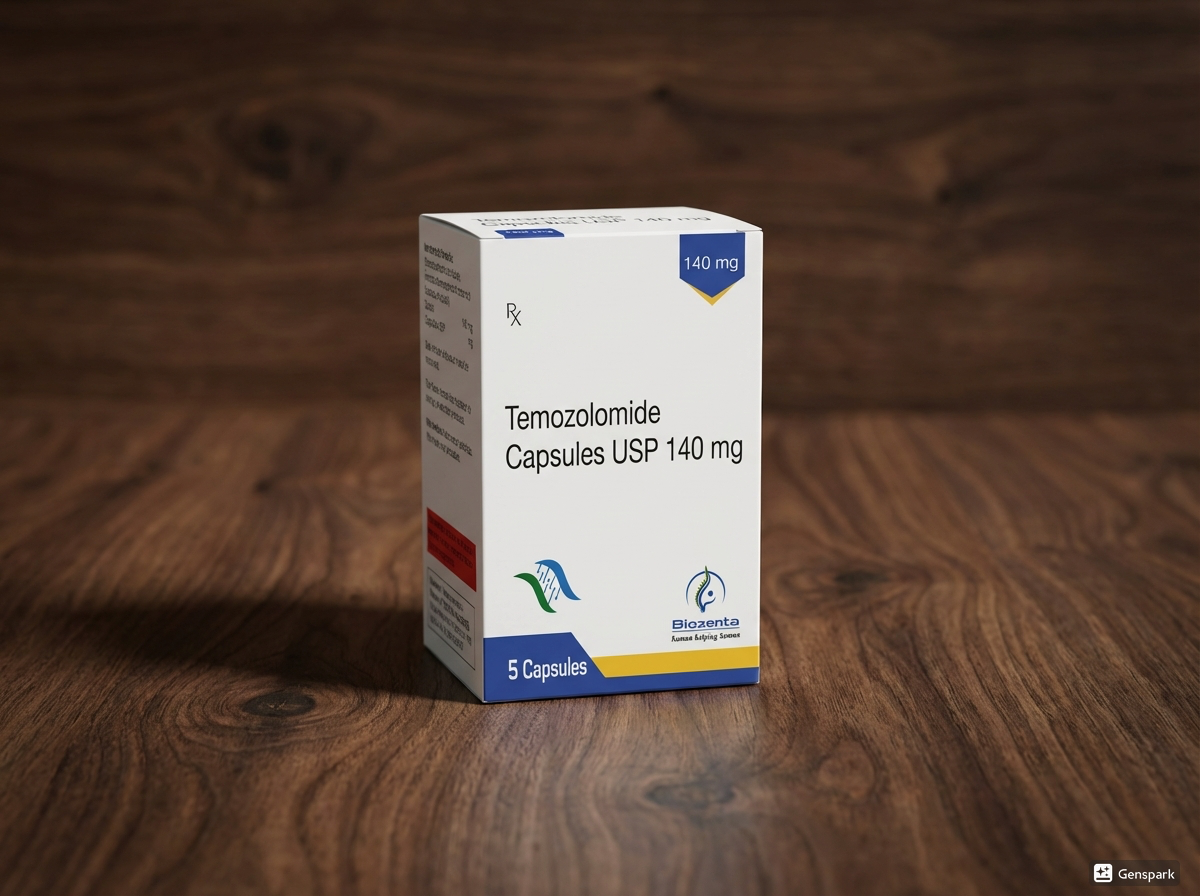
Temozolomide 140 mg Capsules №5
€67,00
Temozolomide 140 mg is a prescription anticancer medication from the class of alkylating agents used to treat malignant brain tumors, including glioblastoma and anaplastic astrocytoma. It works by damaging tumor cell DNA, inhibiting their growth and division. Used only under medical supervision.
Description
Temozolomide 140 mg is a prescription anticancer medicinal product belonging to the class of alkylating agents (cytotoxic chemotherapy drugs). It is widely used in oncology for the treatment of malignant tumors, particularly tumors of the central nervous system. The drug is capable of crossing the blood–brain barrier, making it especially effective in brain tumor therapy.
Pharmacological Class
Alkylating antineoplastic agent (cytostatic).
Mechanism of Action
After oral administration, temozolomide is rapidly converted in the body into its active metabolite, which damages the DNA of tumor cells. This disrupts DNA replication and cell division, ultimately leading to inhibition of tumor growth and cancer cell death. Its ability to penetrate brain tissue makes it particularly effective for primary and recurrent brain tumors.
Indications
Temozolomide is indicated for the treatment of:
-
Newly diagnosed glioblastoma (in combination with radiotherapy, followed by maintenance therapy)
-
Recurrent or progressive glioblastoma
-
Anaplastic astrocytoma
-
Other malignant brain tumors as determined by an oncologist
It may be used as monotherapy or as part of combination therapy.
Dosage Form
Capsules 140 mg
Pack size: 5 capsules
Dosage and Administration
Temozolomide is administered orally according to a treatment schedule prescribed by an oncologist. The dose and duration depend on:
-
Type and stage of tumor
-
Patient’s body surface area
-
Previous treatments
-
Overall clinical condition
Treatment is usually given in cycles, consisting of dosing periods followed by rest periods to allow recovery.
Capsules should be swallowed whole with water, preferably on an empty stomach or as directed by a healthcare professional.
Monitoring During Therapy
Regular medical supervision is essential. Monitoring typically includes:
-
Complete blood count (CBC)
-
Liver and kidney function tests
-
Assessment for signs of infection
-
Evaluation of overall patient condition
Bone marrow suppression is a known risk and requires careful observation.
Possible Side Effects
Common adverse reactions may include:
-
Decreased white blood cells (neutropenia), platelets (thrombocytopenia), or red blood cells (anemia)
-
Nausea and vomiting
-
Fatigue and weakness
-
Headache
-
Increased risk of infections
Serious adverse reactions require immediate medical attention.
Contraindications
-
Hypersensitivity to temozolomide or similar compounds
-
Severe bone marrow suppression
-
Pregnancy and breastfeeding without medical supervision
Drug Interactions
Patients should inform their healthcare provider about all medications being taken, as temozolomide may interact with other drugs, particularly those affecting bone marrow or immune function.
Pregnancy and Fertility
Temozolomide may cause harm to a developing fetus. Effective contraception is recommended during treatment and for a period after therapy as advised by a physician.
Storage and Handling
Store as directed by the manufacturer. Keep out of reach of children. Handle capsules carefully and do not open or crush them.
Prescription Status
Prescription only. Treatment must be initiated and supervised by a qualified oncologist.
Manufacturer
Biozenta Lifescience Pvt. Ltd.
Country of Manufacture
India
Additional information
| Country of origin | |
|---|---|
| Dosage | |
| Volume |


Reviews
There are no reviews yet.