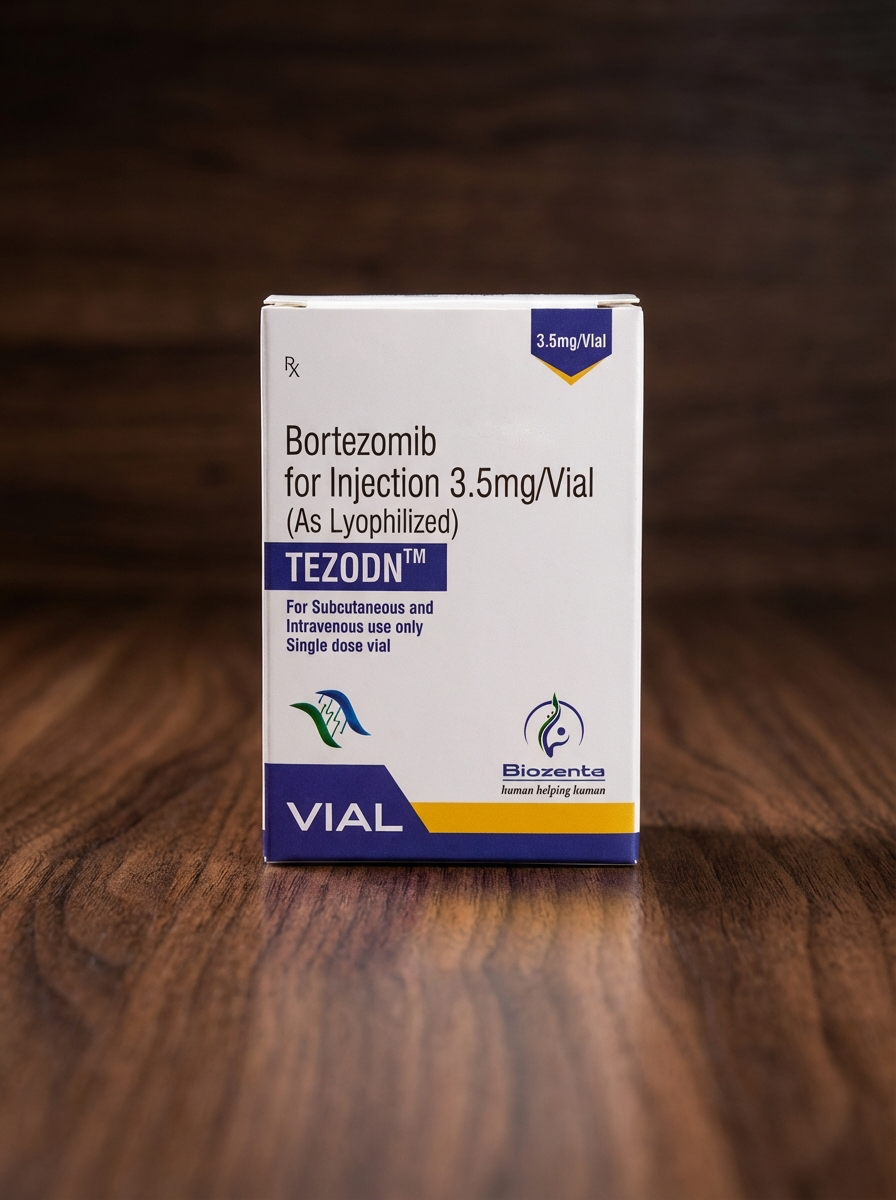
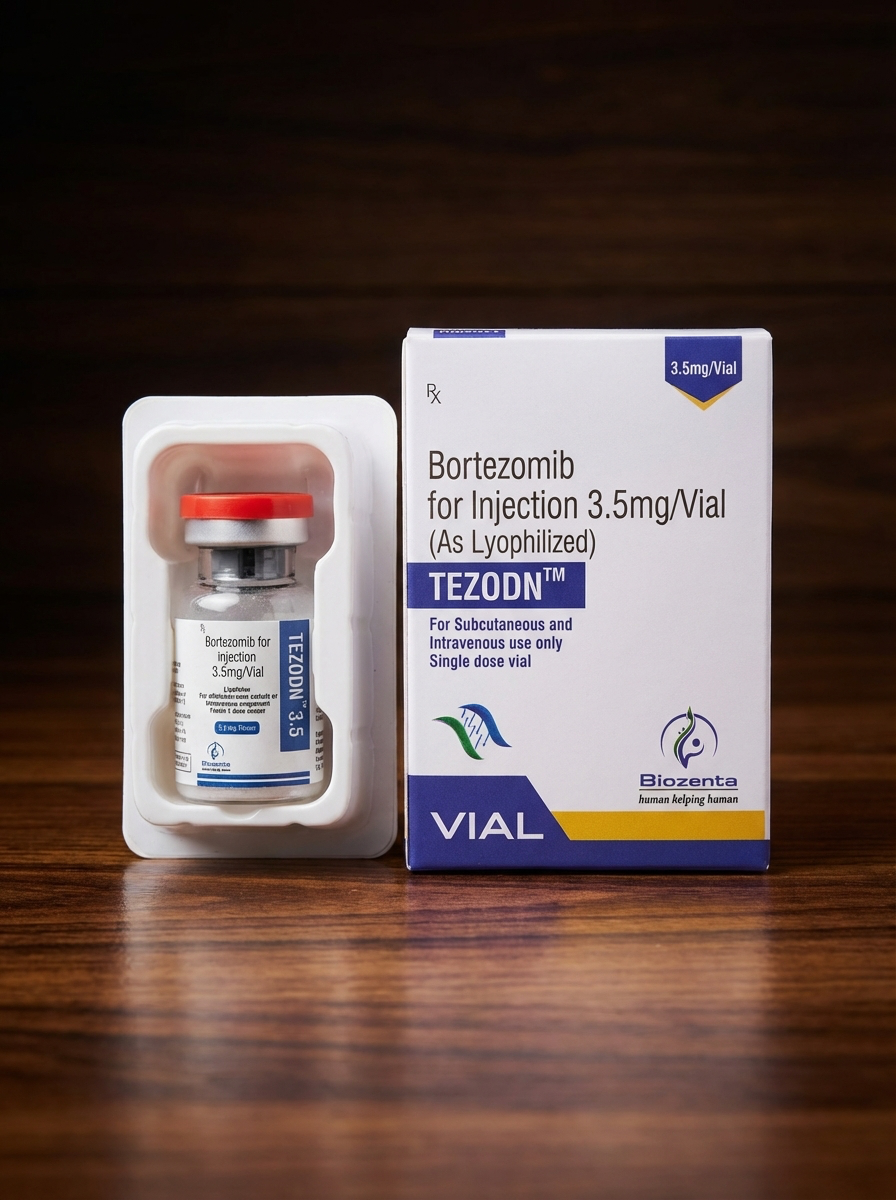
Tezodn (Bortezomib) 3.5 mg/vial, Lyophilized Powder for Injection
€50,00
Tezodn is an antineoplastic medicine containing bortezomib, a proteasome inhibitor used for the treatment of multiple myeloma and mantle cell lymphoma. It is administered intravenously or subcutaneously only under medical supervision.
Description
Tezodn (Bortezomib) 3.5 mg/vial, Lyophilized Powder for Injection
Active ingredient: Bortezomib
Strength: 3.5 mg per vial
Dosage form: Lyophilized powder for solution for injection
Route of administration: Intravenous (IV) or subcutaneous (SC)
Pharmacotherapeutic group: Antineoplastic agents, proteasome inhibitors
Manufacturer: Biozenta Lifesciences Pvt. Ltd.
Country of origin: India
Product Description
Tezodn is a cytotoxic antineoplastic medicinal product containing bortezomib, a selective reversible inhibitor of the 26S proteasome. It is widely used in oncohematology for the treatment of multiple myeloma and mantle cell lymphoma.
Bortezomib disrupts normal protein degradation pathways within malignant cells. By inhibiting the proteasome complex, the drug causes accumulation of abnormal and misfolded proteins, leading to cellular stress, disruption of signaling pathways, and induction of apoptosis (programmed cell death) in cancer cells.
Mechanism of Action
Bortezomib:
• selectively inhibits the 26S proteasome;
• interferes with intracellular protein homeostasis;
• blocks NF-κB signaling pathways involved in tumor cell survival;
• suppresses proliferation of malignant plasma cells;
• induces apoptosis in cancer cells.
The drug demonstrates significant efficacy particularly in plasma cell malignancies.
Indications
Tezodn is prescribed for the treatment of:
• multiple myeloma;
• mantle cell lymphoma;
• other hematologic malignancies according to clinical protocols.
Dosage and Administration
The medication is administered intravenously or subcutaneously by qualified healthcare professionals.
The lyophilized powder must be reconstituted prior to administration according to medical guidelines.
Dosage is calculated individually based on body surface area (BSA), stage of disease, laboratory parameters, and treatment regimen.
⚠ Self-administration is strictly prohibited.
Contraindications
• hypersensitivity to bortezomib or formulation components;
• severe hepatic impairment;
• pregnancy and breastfeeding.
Adverse Reactions
Possible side effects include:
• thrombocytopenia, anemia, neutropenia;
• peripheral neuropathy;
• nausea, vomiting, diarrhea;
• fatigue and weakness;
• fever;
• allergic reactions.
Regular monitoring of blood counts is required during therapy.
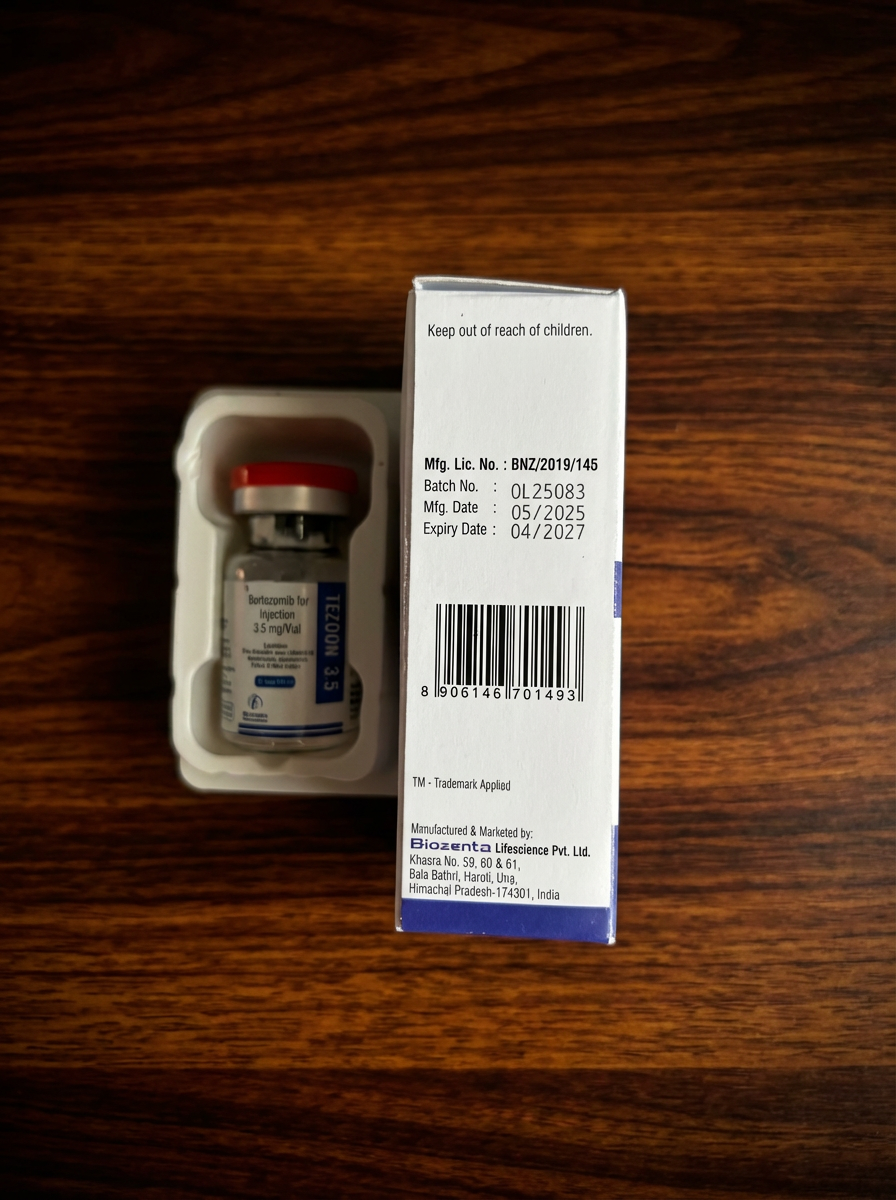
Storage Conditions
Store at temperatures not exceeding 25°C, protected from light, in original packaging.
Keep out of reach of children.
Special Warnings
Treatment must be carried out under the supervision of an oncologist or hematologist.
Baseline and periodic evaluation of liver function and hematological parameters are recommended.

Reviews
There are no reviews yet.